This list may be out of date. We do our best to edit it as we come across news on coverage, but if you see something we have missed, please let us know!
United States
Five augmentation therapies are approved by the U.S. Food and Drug Administration (FDA):
- Prolastin-C® (Grifols)
- Prolastin-C Liquid® (Grifols)
- Aralast NP™ (Takeda)
- Zemaira® (CSL Behring)
- Glassia® (Takeda)
Prolastin has been marketed since 1988 and has an excellent safety record. Aralast NP and Zemaira were introduced to the marketplace in 2003 and Glassia was introduced in 2010. Each was approved by demonstrating that they were comparable to Prolastin in their safety and in augmenting blood and lung alpha-1 levels.
Treatment – Alpha-1 Foundation
TIP: Check with your specific insurance carrierAn Alpha-1 Carrier is a person who has one normal ATT gene (M) and one defective AAT gene (usually S or Z). It does NOT mean you cannot get sick. More as they utilize their own guidelines to make coverage determinations regarding augmentation therapy.
Canada
Glassia is covered through Canadian Blood Services everywhere in Canada, except in Quebec where they use Prolastin for now. Bayshore Pharmacy will provide the product for those in Alberta.
Glassia®, Prolastin-C® and Zemaira® are approved for use in Canada.
Augmentation Therapy – AlphaNet Canada
Glassia can be requested for adult patients (outside of Quebec) with severe A1-PI deficiency and clinical evidence of emphysemaObstructive airway disease in which the walls of the alveoli (air sacs) are damaged or destroyed. More who meet the following criteria*:
- Respirologist has confirmed the diagnosis of severe A1-PI deficiency and clinical evidence of emphysemaObstructive airway disease in which the walls of the alveoli (air sacs) are damaged or destroyed. More and indicated that patient would benefit from treatment with A1-PI product.
- A1-PI deficiency, defined as serum A1-PI levels <11 μmol/L or < 57 mg/dL before start of the treatment.
- Clinical evidence of obstruction (FEV1 <80%).
- Patients must be nonsmokers for at least six months.
- For patients who have not received a lung transplant.
*Criteria are based on recommendations from the Canadian Agency for Drugs and Technologies in Health (CADTH)
Patients should contact their respirologist to determine if Glassia is an appropriate treatment option and whether they meet the required eligibility criteria.
Augmentation Therapy – AlphaNet Canada
TIP: AlphaNet Canada is a great
Europe
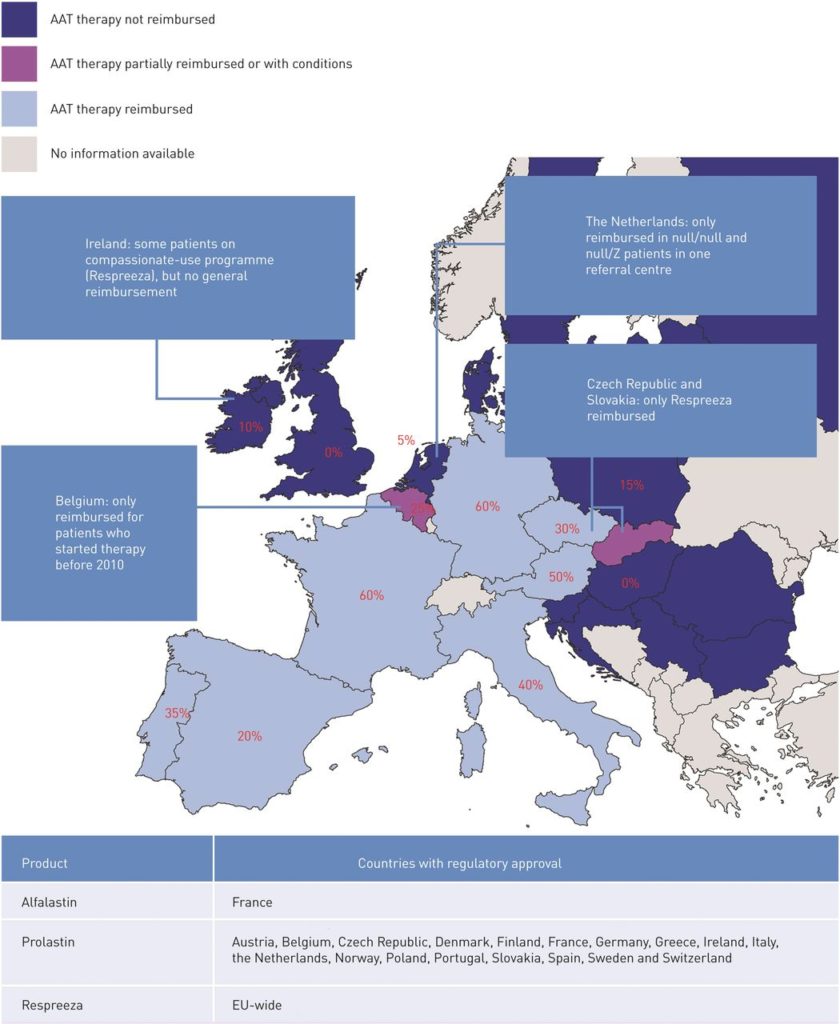
ERJ Open Research 2019
Respreeza and Prolastina were both approved for reimbursement in Denmark January 22nd, 2020.
In Switzerland and Austria, augmentation therapy is reimbursed but in Ireland it is not
RCSI study shows survival benefit of augmentation therapy for people with the genetic lung condition AATalpha-1 antitrypsin MoreD – RCSI
UK
Australia
Not available.
If we haven’t covered your country, and you can find documentation of what is available and how it is covered, please send it to us!

